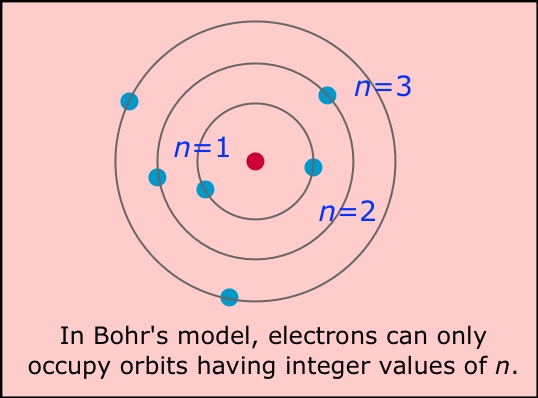
bohr's model consist of four principles
1)Electrons assume only certain orbits around the nucleus. These orbits are stable and called "stationary" orbits.
2)Each orbit has an energy associated with it. For example the orbit closest to the nucleus has an energy E1, the next closest E2 and so on.
3)Light is emitted when an electron jumps from a higher orbit to a lower orbit and absorbed when it jumps from a lower to higher orbit.
4)The energy and frequency of light emitted or absorbed is given by the difference between the two orbit energies,
1)Electrons assume only certain orbits around the nucleus. These orbits are stable and called "stationary" orbits.
2)Each orbit has an energy associated with it. For example the orbit closest to the nucleus has an energy E1, the next closest E2 and so on.
3)Light is emitted when an electron jumps from a higher orbit to a lower orbit and absorbed when it jumps from a lower to higher orbit.
4)The energy and frequency of light emitted or absorbed is given by the difference between the two orbit energies,